|
Greenhouse gas concentrations are measured in parts per million, parts per billion, and even parts per trillion. Larger emissions of greenhouse gases lead to higher concentrations in the atmosphere. How abundant are greenhouse gases in the atmosphere?Ĭoncentration, or abundance, is the amount of a particular gas in the air. With global warming potentials (GWPs) that typically range from thousands to tens of thousands, they are sometimes referred to as high-GWP gases because, for a given amount of mass, they trap substantially more heat than CO 2.Įach gas's effect on climate change depends on three main factors: 
Fluorinated gases are typically emitted in smaller quantities than other greenhouse gases, but they are potent greenhouse gases. Fluorinated gases (especially hydrofluorocarbons) are sometimes used as substitutes for stratospheric ozone-depleting substances (e.g., chlorofluorocarbons, hydrochlorofluorocarbons, and halons). Fluorinated gases: Hydrofluorocarbons, perfluorocarbons, sulfur hexafluoride, and nitrogen trifluoride are synthetic, powerful greenhouse gases that are emitted from a variety of household, commercial, and industrial applications and processes.Nitrous oxide (N 2O): Nitrous oxide is emitted during agricultural, land use, and industrial activities combustion of fossil fuels and solid waste as well as during treatment of wastewater.Methane emissions also result from livestock and other agricultural practices, land use, and by the decay of organic waste in municipal solid waste landfills. Methane (CH 4): Methane is emitted during the production and transport of coal, natural gas, and oil.Carbon dioxide is removed from the atmosphere (or "sequestered") when it is absorbed by plants as part of the biological carbon cycle. Carbon dioxide (CO 2): Carbon dioxide enters the atmosphere through burning fossil fuels (coal, natural gas, and oil), solid waste, trees and other biological materials, and also as a result of certain chemical reactions (e.g., cement production).For more information on the other climate forcers, such as black carbon, please visit the Climate Change Indicators: Climate Forcing page. This section provides information on emissions and removals of the main greenhouse gases to and from the atmosphere. Gases that trap heat in the atmosphere are called greenhouse gases. Greenhouse Gas Emissions and Sinks: 1990–2021. This net sink is not shown in the above diagram. All emission estimates from the Inventory of U.S. Percentages may not add up to 100% due to independent rounding. Land Use, Land-Use Change, and Forestry in the United States is a net sink and offsets 12% of these greenhouse gas emissions. Emissions in 2021 = 6,340 Million Metric Tons of CO₂ equivalent (excludes land sector). The reaction of carbon monoxide and copper oxide will result in the formation of copper and carbon dioxide.Total U.S. What will be the product of carbon monoxide and copper oxide?.The production of carbon monoxide through inadequate combustion of fuels can be described by the given equation: It means the reaction of steam with carbon or its source releases carbon monoxide and hydrogen. The cause of the production of carbon monoxide is the inadequate combustion of fuels. During the water gas shift process, carbon monoxide is used to produce hydrogen.It is used as a reagent during the production of aldehydes.It also plays an important role in infrared lasers.With the help of carbon monoxide, the rusted layer of metal can be removed easily.To acidify certain food products like jam and cola, carbon monoxide is used.Carbon monoxide is a major component of the production of methanol. 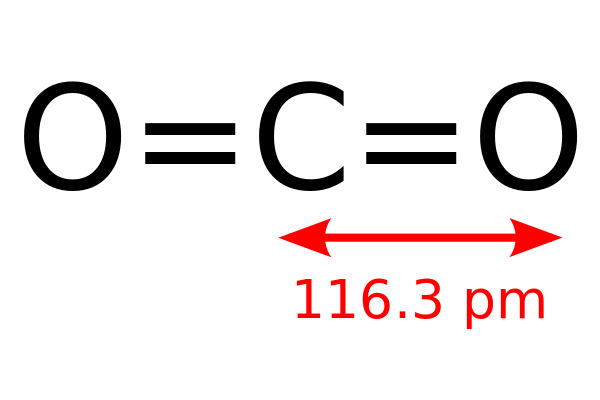
It also plays an important role in metallurgical applications.One of the most important uses of Carbon monoxide is manufacturing synthetic chemicals.The bond length between the atoms of oxygen and carbon is about 112.8 pm. Carbon monoxide has extensive use, including producing useful compounds such as fragrances, drugs, and even fuels.Ĭarbon Monoxide comprises a single atom of each carbon and oxygen, bound by a triple bond, including one sigma bond and two pi bonds. One of the major biological sources responsible for carbon monoxide production in humans. The most common source of Carbon monoxide is thermal combustion, although several other biological sources contribute to the generation and emission of carbon monoxide. Carbon monoxide forms a useful ligand called carbonyl, which is used as a major ingredient in several chemical industries.Ĭarbon monoxide comprises each atom of carbon and oxygen its chemical formula is given as CO. Carbon monoxide belongs to the oxocarbon family, and it is one of the simplest molecules in its family. It comprises one oxygen atom and one carbon atom, which are combined by a triple bond. Carbon monoxide is a flammable, tasteless, odorless, and colorless gas, which is quite less heavy than air.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
